The number of irreversible reactions used in the formation of synthetically useful covalent bonds largely outweighs that of reversible ones. Yet, the last period has witnessed a renewal of interest in the use of reversible reactions for synthetic purpose thanks to the birth of Dynamic Combinatorial Chemistry (DCC) mainly due to J.-M. Lehn, J. K. M. Sanders and S. Otto. In particular DCC has the potential to be a powerful tool for the synthesis of macrocyclic species under thermodynamic control because efficient cyclic receptors may be selected among a family of interconverting linear and cyclic members of a dynamic library (DL) upon the addition of a suitable template (T), via repeatedly occurring bond dissociation-recombination processes. Some reversible reactions such as acetal exchange, transimination and imine and olefin metathesis have been studied in our lab, in the frame of DCC. Furthermore, we are strongly interested in the physico-chemical laws governing the macrocyclization reactions that are very often at the basis of the systems studied in DCC. In the last period we have been working on a re-elaboration of the Jacobson-Stockmayer theory, which includes knots and mechanical bonded species such as catenanes, into the classical treatment, and on experimental results proving our theories.
Related representative publications:
S. Albano, G. Olivo, L. Mandolini, C. Massera, F. Ugozzoli, S. Di Stefano, Journal of Organic Chemistry 2017, 82, 3820−3825.
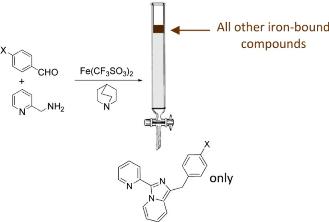
S. Di Stefano, G. Ercolani, Journal of Physical Chemistry B 2017, 112, 649–656.
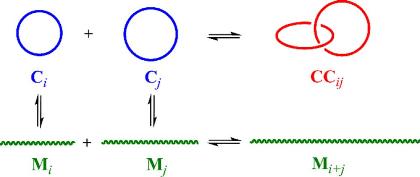
S. Di Stefano, G. Ercolani, Advances in Physical Organic Chemistry 2016, 50, 1–76.
J. A. Berrocal, L. M. Pitet, M. M. L. Nieuwenhuizen, L. Mandolini, E. W. Meijer, S. Di Stefano, Macromolecules 2015, 48, 1358–1363.
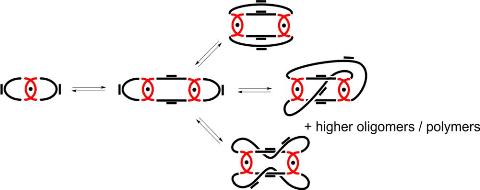
M. Ciaccia, S. Di Stefano, Organic & Biomolecular Chemistry 2015, 13, 646–654.
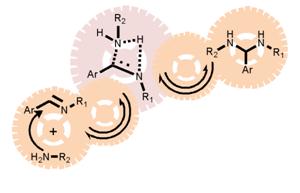
Updated mechanisms operating in imine chemistry in organic solvents are reviwed and critically discussed.
M. Ciaccia, I. Tosi, L. Baldini, R. Cacciapaglia, L. Mandolini, S. Di Stefano, C. A. Hunter, Chemical Science 2015, 6, 144–151.
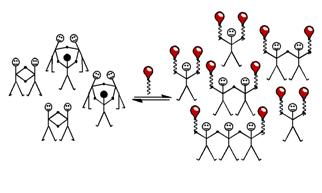
Addition of chain‑stoppers to diluted Dynamic Libraries allows determination of EM of macrocycles.